AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Co2 molecular geometry9/24/2023 
Furthermore, the polymers in D2O collapse at higher temperatures due to decreasing solubility of the side chains in water. Understanding the molecular structure of a compound can help determine the polarity, reactivity, phase of matter, color, magnetism, as well as the biological activity. The volume of the cylinder shows no concentration dependence. Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. % solution in D2O, the polymers are partially contracted at room temperature and their conformation can be described by the form factor of a rigid cylinder. % solution in d-toluene the polymers assume wormlike chains and gradually contract with increasing polymer concentration. The polymer conformation was studied in two solvents, d-toluene and D2O, with the aim of understanding the influence of solvent/polymer more » interactions on the resulting structures. The hydrophobic backbone and hydrophilic side chains interact differently with solvents depending on their polarity, which makes the conformation very sensitive to the solvent quality. The homopolymers consist of a polynorbornene (PNB) backbone with a degree of polymerization (DP) of 50, and each backbone monomer has a grafted Ethylene Glycol (EG) side chain with an average DP of 6.6. The structure of thermo sensitive poly(methoxyoligo(ethylene glycol) norbornenyl esters) homopolymers in dilute solution was investigated by Small Angle Neutron Scattering (SANS). Results from the TPES indicate that the molecular geometry is distorted upon ionization, that production of BFlowered 3, raised + occurs with low. PDMS 550 absorbs more H 2 than all of the other solvents. PDMS 550 is a very good CO 2 solvent, absorbing more CO 2 than all of the other solvents at all temperatures except for PEGDME 250 at 25 ☌. Even though the two dipole moments are equal in magnitude (since they are both oxygen hydrogen bonds), they are not arranged symetrically about the central atom and thus will not cancel out.=. 
Carbon dioxide will not be polar because both dipole moments are equal in magnitude (since they are both carbon oxygen bonds) and arranges symetrically about the central atom in a linear geometry. The molecular geometry of any compound can be determined by the VSEPR theory. Water is a bent molecule with two polar bonds. 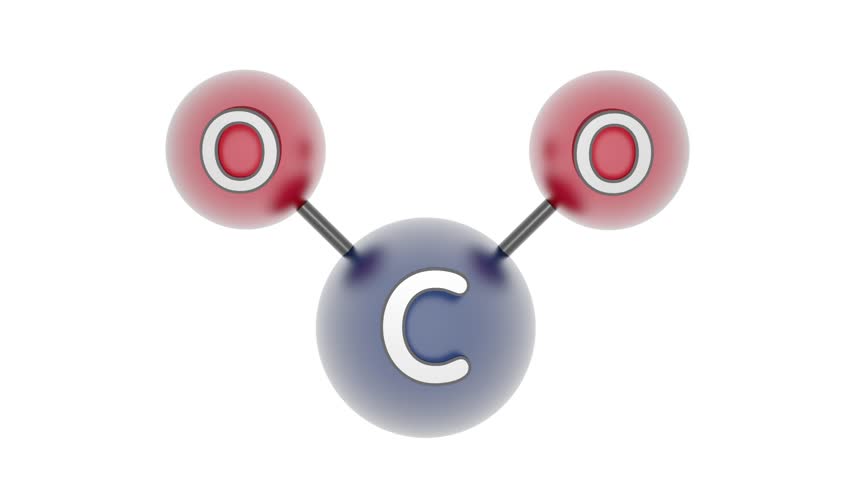
Carbon dioxide is a linear molecule with two polar bonds. Let's look at two examples: carbon dioxide and water. In the following geometries, the atoms are arranged symetrically about the central atom: linear, triangular planar, tetrahedral, square planar, triangular bipyrimidal, and octahedral. They are symetrically arranged about the central atom. the bonds are made with the same element) ANDĢ. Any resulting dipole indicates a polar molecule.ġ. Step 2: Using the molecular geometry, determine if any of the dipole moments will cancel. You have found all of the polar bonds in each of these three molecules. Lewis structures for H 2S, BF 3, and CCl 2H 2Īre shown below. This occurs because of a difference in electronegativity of the two atoms that share the electrons. Remember that a polar bond is one in which the electrons are unevenly distributed. Step 1: Indicate polar bonds in molecule or ion. As you work through these steps you will see that molecules with polar bonds are not necessarily polar molecules. There is a series of steps you can take to determine if a molecule is polar or not. 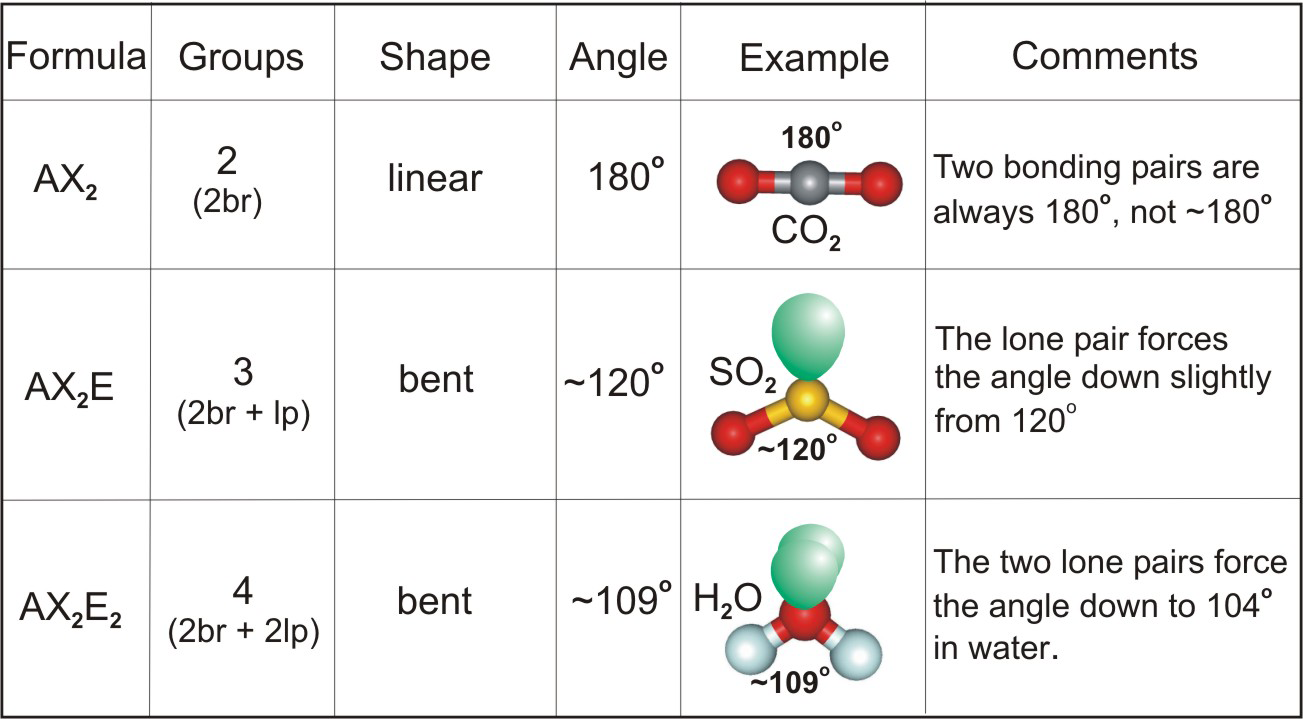
Likewise molecules in which there is an accumulation of electron density at one end of the molecule, giving that end a partial negative charge and the other a partial positive charge, are called polar molecules. The electron density of a polar bond accumulates towards one end of the bond, causing that end to carry a slight negative charge and the other end a slight positive charge.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
